Types of patent applications granted in India
- Ordinary or Non-Provisional Application
- Convention Application
- PCT International Application
- PCT National Phase Application
- Patent of Addition
- Divisional Application
1. Ordinary or Non-Provisional Application
Ordinary or Non-Provisional application is filed if the applicant doesn't have any priority to claim or in a case where the application is not filed in pursuance of any preceding convention application. Non0 provisional application, when filed, must be supported by a complete specification depicting the invention in detail.
2. Convention Application
A convention application is filed for claiming a priority date based on the same or substantially similar application filed in any of the convention countries. The applicant is required to file an application in the Indian Patent Office within a year from the date of the initial filing of a similar application in the convention country.
3. PCT International Application
A PCT Application is an international application to streamline patent application process in many countries at one go. It is governed by the Patent Corporation Treaty and can be validated in up to 142 countries.
4. PCT National Phase Application
A national phase application is filed in each of the country wherein the protection is sought. The national phase application must be filed within 30 or 31 months from the priority date or the international filing date, whichever is earlier.
5. Patent of Addition
The applicant can file a patent of addition if there is a modification of the invention which has already been applied for or patented. A patent of addition can only be granted after the grant of the parent patent; hence, no separate renewal fee is to be remitted during the term of the main patent.
6. Divisional Application
Divisional application is filed if the applicant wishes to divide an application to furnish two or more applications if a particular application claims for more than one invention. The priority date for divisional application is similar to that of the parent application.
Criteria for a patent to be granted
- Novelty: According to Section 2(1)(j) of the Indian Patent Act, "invention" is a new product or process involving an inventive step and capable of industrial application. Further, Section 2(l) of the Act defines 'new invention' as any invention or technology which has not been anticipated by publication in any document or used in the country or elsewhere in the world before the date of filing of patent application with complete specification, i.e., the subject matter has not fallen in public domain or that it does not form part of the state of the art.
- Inventive Step: According to Section 2(1) (ja) "inventive step" means a feature of an invention that involves technical advance as compared to the existing knowledge or having economic significance or both and that makes the invention not obvious to a person skilled in the art.
- Industrial Application: The third criterion of patentability is that the invention should be capable of industrial application. Section 2(1)(ac) of the Patents Act, 1970, defines "Capable of Industrial application" in relation to an invention as the invention is "capable of being made or used in an industry." Therefore, to be patentable, an invention must be useful.
Basis for rejection of patents in India1
The rejections by the IPO are raised mostly in section 15 proceedings, which accounts for 1637 rejections. Section 15 is not quoted in isolation and is often used in conjunction with other sections, at times, the IPO calls for a hearing (under section 14), requesting the applicant for clarifications. In cases where the applicant fails to clarify the objections raised under section 14, the Controller rejects the application under section 15. In 54 cases, the Controller rejected the application solely based on section 16, i.e. the application did not qualify as a divisional application under the Act. Section 16 was otherwise quoted along with other sections in 117 cases.
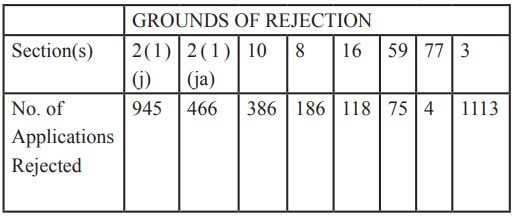
Further, as illustrated in the table above, section 3 was used in 65% of cases either singly or in combination with other sections to reject the patent applications. Out of the total number of 1723 applications, 1113 applications were rejected by citing section 3 as an objection.
Section 3 has many sub-sections out of which section 3(d) that deals with patentability of known substances was used, either alone or in combination with other sections in 771 cases (69%). In combination with other subsections like section 3(e) and section 3(i), section 3(d) was used in 36% of cases where an objection of section 3 was raised.
REJECTION OVERVIEW: PHARMA VS OTHERS2
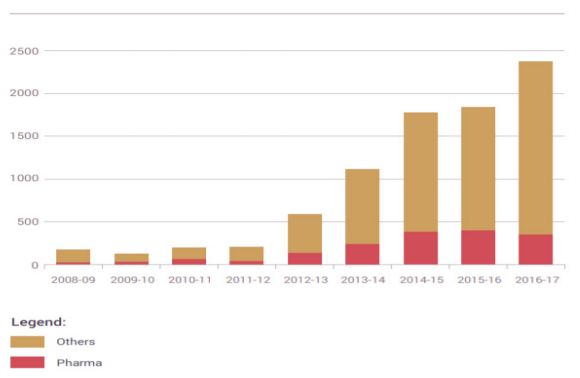
It is evident from the graph below that rejection of pharma were reduced as compared to the non-pharma patents, wherein the rejections were increased.
As illustrated in the bar graph above, there was an increase in rejection of pharma patents from the year 2013-2014 to 2014-2015. However, the number of rejections of the same remained constant there through. On the contrary, there was an increase in the rejection of other patents as compared to the pharma patents.
Footnotes
1 http://accessibsa.org/media/2017/12/Rejected-in-India.pdf
2. http://accessibsa.org/media/2017/12/Rejected-in-India.pdf
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.


