Britain is a large player in the global genomics field. With a rich history of scientific discoveries, the genomics sector in the UK continues to grow. In this article, we take a look at how and where innovation and growth is occurring for the British genomics industry, and take a deeper look into 10 exciting businesses that are taking the sector by storm.
Investment in British genomics
Genomics is the study of the complete set of DNA in a person or an organism. As almost every cell in the human body contains a complete copy of the genome, which contains all the information needed for development and growth, studying genomes may help with understanding how genes interact with one another and the environment, and how certain diseases (such as cancer, diabetes, and heart disease) form. This, in turn, can lead to the development of new ways to diagnose, treat, and prevent diseases like these.
From the discovery of the double helix structure of DNA by British scientists studying at King's College in the 1950's, to Fred Sanger's development of DNA sequencing in the 1970's, the UK has long been viewed as a leader in the world of genomics development. With developments in the sector continue to gather pace, we take a look into the dynamic world of the UK genomics sector and highlight 10 innovative and interesting UK companies that are contributing to its commercial development.
Besides historic scientific discoveries in the field, the UK's leading position in genomics can be contributed to a number of factors. In no particular order, the UK offers particularly attractive tax reliefs to innovative businesses such as R&D tax credits, the Enterprise, and Seed Enterprise Investment Schemes (EIS and SEIS respectively). These reliefs make the UK a particularly attractive location for talented scientists looking to research and develop innovative drugs and technologies in the space. Secondly, the contributions of the NHS cannot be forgotten; the NHS provide the ability to link a lifetime of medical records with a person's genome data on a large scale, which furthers the understanding of diseases and provides insight into the complex relationship between genes, the surrounding environment, and illness. Cementing the UK's position as the world leader in genomics are various initiatives including the following:
- 100,000 Genome Project's seeking to sequence and study the role of genes in health and disease,
- The UK Biobank, a large-scale biomedical database and research resource, and
- The NHS Genomics Medicine Service, who are driving to deliver a single national testing directory covering use of all genomic technologies, and to build a national genomic knowledge base that will provide real world data to inform both academic and industry R&D.
In order to understand how the UK is driving innovation and commercial growth in the genomics field, we used data from Beauhurst to understand the landscape of the sector and understand where investment is taking place.
Characteristics and fundraising activity of genomic businesses
*Beauhurst
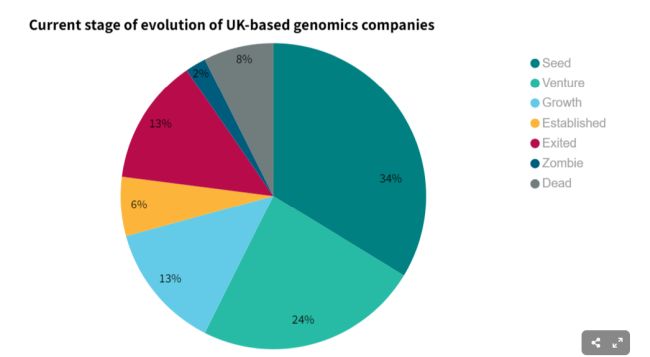
Genomics, as a sector, is still in its relative infancy. Of the 284 genomic companies on Beauhurst, the majority, at 58%, are in the early stages of development. This is corroborated by over half, at 63%, being incorporated within the last 10 years.
Of 909 fundraisings, where the investment amount is known, £6bn has been invested into genomics over the past 20 years, with an average cheque size per company of £6.89m. A stand-out year for genomics, in terms of fundraising, was 2021, with £1.44bn being raised in total during that one year alone. Average cheque size per company was also up during that time, to £15.8m. The total amount being invested in this space increased by 98% in 2021 from 2020, however, in 2022, investment decreased by 54% as the wider equity investment market saw significant drops in appetite and activity.
Of the 284 companies we accessed data for, we found that 36 have exited with the majority, at 92%, still actively trading. Over half (53%) of exits occurred between 2021 and 2023, with 28% of these exiting via Initial Public Offering (IPO), achieving market capitalisations ranging from £18.1m to £3.4bn. The remaining 72% of exits were private acquisitions, with comparatively modest acquisition prices ranging from £9.2m to £588m.
Sector breakdown of genomics businesses
*Beauhurst
*Percentages are of company sample
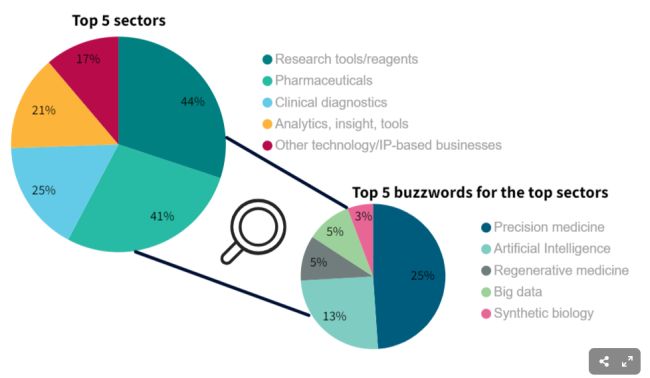
The top 5 sectors are not all that surprising; they are what is expected given the nature of the work that is conducted in the genomics sector. Research tools/reagents and pharmaceuticals seem to both be big focuses of the sectors (at 44% and 41%, respectively). By looking at the top buzzwords for these sectors, we can see if there are any additional focuses for genomics businesses that would not be captured otherwise. As we can see, precision medicine (25%) and AI (13%) are the most prominent buzzwords.
Due to the popularity of research tools/reagents and pharmaceuticals, these two appear consistently when looking at the success of the different sectors. For companies that exited through an IPO, research tools/reagents average market capitalisation is 115% above that of pharmaceuticals, despite the same sample sizes. This is however switched for acquisitions, as pharmaceuticals have an 80% higher average acquisition price than research tools/reagents, with half the number of companies. In terms of the cheque size for their latest fundraising, the average is the same for both of these sectors at £50m, despite research tools/reagents having a bigger sample size.
Company profiles
We now place the spotlight on 10 exciting genomics businesses.
DnaNudge
DnaNudge, a London-based Imperial College London spinout, was founded in 2015 with the mission to democratise healthcare. Its founders are Professor Chris Toumazou, a multi-award-winning innovator, London's first Regius (Royal) Professor of Engineering and founder of the Imperial College Institute of Biomedical Engineering, and Dr Maria Karvela, a published biologist, geneticist, and a leukaemia researcher.
DnaNudge have created a rapid out-of-lab genotyping technology that is able to decode aspects of DNA, called Single Nucleotide Polymorphisms (SNPs), in 90 minutes. This information is then used to provide guidance to healthier everyday choices, in order to help improve nutrition and skincare. The process begins by inserting a user's swab into their patented DnaCartridge. The sample of the DNA is then rapidly heated and cooled, as many times as needed, in their NudgeBox, a portable Polymerase chain reaction (PCR) machine, in order to make the sample big enough for detection on their microchip. Once this process is completed, the switch is made onto their chip. As the DnaCartridge contains a multiplex array of 72 wells preloaded with well-researched marker strands of DNA, if the inserted DNA matches any of the markers, a chemical reaction will take place that activates the chip. The NudgeBox then detects these activations, and, in 90 minutes, the results are sent directly to the individual's smartphone. The sample is then destroyed, to ensure full confidentiality.
With the use of the DnaBean accessory or a smartphone, users are able to scan products to see if said product is red or green. Green means that the product is a good choice, with red meaning the converse. When a red product is scanned, green alternatives will be offered, allowing users to make the best daily choices for them, based on their DNA.
2023 has been an interesting year for DnaNudge. In March, they announced their strategic partnership with Mylab Discovery Solutions, India's leading biotech company, with the mission to develop next-generation molecular diagnostics for diabetic foot infections and other key health conditions. The first output of this partnership is a diabetic foot ulcer detection kit, assisting clinicians in rapidly identifying bacteria involved in infections and allowing optimal treatment for patients at the point of decision, ultimately reducing the risks of acute complications, limb amputation and antimicrobial resistance.
They have also been selected by WIRED Consulting as a 2023 WIRED Trailblazer, where co-founder Dr Maria Karvela joined an exclusive meeting of visionary leaders from fast-growth London businesses to discuss their technology and innovation story, and explored how innovative companies like theirs are driving positive outcomes for both people and the planet. In addition, in November, they launched a revolutionary genomic handheld device with NantNudge, the first handheld polymerase chain reaction (PCR) genomic diagnostic cartridge and chip. This device will transform real-time decision support for healthy lifestyle choices. It also has the possibility of transforming the diagnosis of infections and cancer risk, which are based on personal genomics at point-of-decision.
Ochre Bio
Founded in 2019 and based in Oxford, Ochre Bio are committed to making healthy livers available for patients everywhere. The co-founders are Jack O' Meara (CEO), a biochemical engineer who is now an entrepreneur, and Quinn Wills (CSO), a MD/PhD who also has degrees in genomics, mathematics, and statistical/machine learning. They are a biotechnology company with a mission of developing therapies for important liver health challenges, such as increasing donor liver supply and reducing cirrhosis complications, by utilising a combination of genomic deep phenotyping, precision Ribonucleic acid (RNA) medicine, and testing in live human donor livers.
Ochre Bio are developing therapies for the biggest liver health challenges, including:
- Deep phenotyping livers at scale,
- Designing precision RNA therapies to reprogram them, and
- Regenerating human livers outside the body.
They conduct their research with the use of donor livers that are not suitable for organ donation but are suitable to use for drug research. They keep the livers 'alive' for days at a time by using machines that mimic physiological conditions. The liver data is analysed by using machine learning, so that targets for its drugs are able to be identified and validated.
A notable event for Ochre Bio is when, during October of 2022, they raised $30m in Series A funding. This funding is for supporting the development of RNA therapies for chronic liver disease and for identifying a drug candidate to test in real live humans.
Touchlight

Johnny Olson, now the Executive Chairman, founded Touchlight, the developer of a novel, synthetic DNA vector (dbDNA") and an enzymatic manufacturing process, in 2007. He was inspired by the advent of genome sequencing as he believed that this would be a driving force for innovation in genetics, foreseeing DNA underpinning the next generation of medicines. With the current methods of creating DNA, at the time, having inherent limitations in speed, safety, cost, capacity and scalability, there was a clear need for a new innovate method of DNA production that would be well-matched for the growing requirements of this rapidly expanding sector. Therefore, at the time of their establishment, Touchlight's main mission was to make DNA to power the genetic medicine revolution.
Their doggybone DNA (dbDNATM) is a novel, synthetic DNA vector whose enzymatic production allows Touchlight to create DNA at the speed, scale and purity needed to assist the rapid growth of genetic medicines. This dbDNATM is named after its schematic structure, a minimal, linear, double stranded, and covalently closed DNA structure. It is able to encode long, complex, or even unstable DNA sequences, as well as eliminating bacterial sequences and has a strong expression profile.
In April 2023, they announced a collaboration with 20Med Therapeutics, a leader in non-viral delivery and development of gene therapies and vaccines, which will work to establish a novel vaccination platform, combining Touchlight's rapid enzymatic doggybone DNA platform with their own bioresponsive polymer nanoparticle technology.
Only a month later, in May 2023, Touchlight officially announced the completion of the redevelopment and expansion of their UK manufacturing facility on the banks of the River Thames. Its manufacturing capacity has tripled, now capable of producing more than 8kg a year, exceeding the current growing global supply of plasmid DNA for genetic medicine. The facility now encompasses 11 suites and two class B filing suites with class A filing cabinets, warehousing, and a quality control laboratory capability. They have also already received a successful customer quality audit from a Big Pharma company.
During July, Touchlight announced the launch of a Discovery grade dbDNA" (enzymatic doggybone DNA) that will offer support for customer candidate screening, including allowing customers to receive fast and easy access to 0.5 to 1mg of Research Use Only (RUO) grade dbDNA, with them being able to use it in as soon as 4 weeks from receipt of template DNA.
PhoreMost

PhoreMost, a spinout of the University of Cambridge's MRC Cancer Unit who are driving the discovery of tomorrow's medicines, was founded in 2014 by Dr Chris Torrance (Chair, and former-CEO), Professor Ashok Venkitaraman and Dr Grahame Mckenzie. Dr Torrance is a cancer researcher and entrepreneur, with him founding Horizon Discovery in 2007 with the main mission of translating advances in human genome editing into a range of research tools and services in order to assist the discovery of new and improved 'Personalized Medicines'. Professor Venkitaraman is the Director of the Cancer Science Institute of Singapore, a Distinguished Professor of Medicine at the National University of Singapore, and Program Director at A*STAR. Dr Mckenzie, the former CSO of PhoreMost, is now the VP Discovery Biology at Mosaic Therapeutics.
PhoreMost are the developers of SITESEEKERTM, a next-generation phenotypic screening platform that is based on the company's PROTEINiTM technology, which is able to recognise the best new targets for future therapies as well as how to drug them. This platform has the chance to greatly increase the diversity of novel therapeutics for cancers and other diseases of unmet need. Across the entire human proteome, this platform is also able to systematically unmask druggable sites and directly link them to useful therapeutic functions in a live-cell context. PhoreMost has a unique approach to developing new life-changing therapies through a combination of this platform, and the exploitation of the concurrent revolutions of deep learning in drug design and protein structural simulation.
At the beginning of 2023, PhoreMost triggered a milestone payment in their multi-project target discovery collaboration with Boehringer Ingelheim, one of the world's largest pharmaceutical companies. This collaboration started in January 2020, with the deployment of PhoreMost's in-house expertise and SITESEEKERTM platform towards disease-relevant pathways, which were selected by Boehringer Ingelheim. The trigger for this milestone payment was the identification and validation of novel targets, in support of Boehringer Ingelheim's mission to develop first-in-class therapies that can transform patients' lives.
Evox Therapeutics

Evox Therapeutics, an Oxford-based biotechnology company focusing on improving the natural delivery capabilities of exosomes, was co-founded in 2016 by Dr. Per Lundin, Professor Matthew Wood, and Dr. Samir El Andaloussi. Dr. Lundin (Chief Business Officer) also serves as the non-executive director of Combigene (an AAV gene therapy company) and is a board director of Juvenile Diabetes Research Foundation UK. Professor Wood is a Professor of Neuroscience and the Deputy Head of the Medical Sciences Division at Oxford University. Dr. El Andaloussi is an Associate Professor of Biomolecular Medicine at the Karolinska Institutet and focuses his research on exosome-based vehicles for macromolecular drug delivery.
Evox Therapeutics' mission is to create novel exosome-based therapeutics able to treat rare and severe diseases that have limited treatment options, with the overall goal to positively impact human health. Some of the limitations of proteins, antibody, and nucleic acid-based therapies could be addressed by exosome-based drugs due to their ability to deliver to cells and tissues out-of-reach of other drug delivery technologies. Evox Therapeutics are leaders in the development within this emerging space, with the company aiming to make new breakthrough and discoveries in the treatment of severe and rare genetic disorders.
They are combining exosomes' delivery capabilities with targeting technology and sophisticated biomolecular engineering approaches. One such example of this is DeliverEXTM, their proprietary technology platform, which allows modification of exosomes, a way to load drugs into exosomes, and targeting the delivery of said exosomes to organs of interest. They have had success in delivering drug-loaded exosomes into target organs, including the central nervous system and other intractable tissues.
In May 2023, they were granted a US patent that covers a downstream process, utilising ultrafiltration, and size-based separation steps, for purifying engineered exosome, and importantly it protects a process commonly used in the exosome therapeutics field. Alongside the other four manufacturing patents issued to the company in 2022, this patent helps to build upon Evox Therapeutics' manufacturing patent protection, now covering key purification and loading methods in key jurisdictions.
Further in June, they announced that they purchased Codiak Biosciences engEx-AAV" technology platform, along with all of the intellectual property rights and the secured exclusive access to certain associated engineering and manufacturing rights. This technology platform allows for exomes to be active loaded by adeno-associated virus (AAV) followed by their release when said exosomes are delivered into recipient cells. This improves AAV delivery, increases the breadth of expression in pre-clinical models, and shields AAVs from neutralising antibodies.
Also, during this year, they revealed a research collaboration and option agreement with the Icahn School of Medicine at Mount Sinai, which will have the aim of developing exosome-encapsulated AAV vectors as a novel gene delivery technology, improving treatment for heart disease.
Sphere Fluidics

A University of Cambridge spinout, Sphere Fluidics is a developer of platforms that have picodroplets at their centre. They were co-founded in 2010 by Frank F. Craig, PhD, MBA; Professor Wilhelm Huck, PhD; and Chris Abell. Frank Craig is the CEO and Board Director as well as the co-founder and a VP of Aurora Biosciences. Professor Huck is one of the Scientific Advisors and a Professor at the Institute for Molecules and Materials at Radboud University, and Chris Abell is the Pro-Vice-Chancellor for Research and Professor of Biological Chemistry at the University of Cambridge.
As mentioned, Sphere Fluidics utilise picodroplets; tiny droplets of aqueous fluid in an oil carrier fluid, known as a single emulsion, or an aqueous core surrounded by an oil shell in an aqueous carrier fluid, known as a double emulsion, with each picodroplet able to encapsulate a cell. Through the careful design of the emulsions' chemistry, biocompatible droplets with minimal leakage of molecules, including small molecules such as metabolites, from the droplet are provided. This produces an ultra-miniaturized test tube that contains media, reagents, and cells, allowing for large-scale experiments to take place at the single cell level.
These picrodroplets can be used for a variety of applications, including antibody discovery, cell line development, cell engineering, cell therapy, and synthetic biology. Two examples of how Sphere Fluidics are utilising technology is, firstly, their Cyto-MineTM, which is a fully automated industrial platform that is able to simplify and accelerate biopharmaceutical discovery and cell line development workflows through its provision of automated, accurate, cost-effective high-throughput screening with gentle processing and reduced contamination. The second example is their Pico-MineTM, a semi-automated platform made to support earlier-stage research with ultra-flexibility for workflows and assays to help discover the valuable and rare biological variants among large cell populations.
During 2023, they celebrated the opening of their new laboratory space, an expansion of their Granta Park base, so that it is now 30k square feet with the inclusion of a demonstration lab and microfabrication facilities. Their technology has also been adopted by third-party companies during this year; their Cyto-MineTM was adopted by Portugal-based FairJourney Biologics to streamline and accelerate customer workflows for the launch of their latest cell line development services, and their Pico-MineTM was chosen by Biosyntia so that its ultra-high-throughput, picodroplet-based screening capabilities could be harnessed to accelerate development of novel industrially-relevant cell factories along with advancing bio-based production of nutritional ingredients.
Plasticell

Starting their journey in 2002, biotechnology company Plasticell is developing novel therapies with the use of precise manipulation of stem cells and more differentiated cell types, by utilising award-winning combinatorial screening technology. Based in Stevenage, this company was founded by Dr Yen Choo, holder of a PhD in Molecular Biology from the University of Cambridge, and the Chairman of Plasticell and of Progenitor Labs. He was also the founder and Chief Scientist of a biotech company Gendaq, a developer of the zinc finger technology which is now used for gene editing. Prior to his entrepreneurial career, he was a staff scientist at the MRC Laboratory of Molecular Biology, with this being where he was a pioneer for the engineering of protein-DNA interactions using combinatorial methods under Nobel laureate Sir Aaron Klug. Alongside his role at Plasticell, he sits on the UK Stem Cell Steering Committee, the overseers of national stem cell research and the UK Stem Cell Bank.
Their mission is to develop products for cancer and rare disease indications by manipulating pluripotent and haematopoietic cells. They are progressing several therapeutic programs, such as expanding hematopoietic stem cells from bone marrow and cord blood, and developing allogeneic immunotherapies, red blood cells and platelets from induced pluripotent stem cells (iPSCs).
Plasticell's CombiCultTM is their award-winning combinatorial screening technology. It allows for the simultaneous testing of thousands of combinations of cell culture variables for the identification of optimal laboratory protocols for any possible outcome in cell biology. This provides the ability to rapidly develop new and improved cell culture protocols, production of human cells for drug screening, cell expansion and differentiation, gene transduction, and the production of protein. Its compatibility with all cell types, stem cells and iPSCs included, allows for it to be deployed in applications such as stem cell expansion and differentiations, gene transduction, optimal cell line selection, and in providing rare human cells for use in vitro drug discovery, toxicology, or clinical trial stratification.
During the first half of 2023, they announced their collaboration with LambdaGen, a Singapore-based biotechnology company who are commercialising LIGIT (Lambda (λ)-Integrase Genome Insertion Tool), a proprietary genome engineering system which allows for large, multi-gene cassettes or DNA fragments to be specifically inserted in the human genome at safe harbour sites. The focus of this partnership is to use genome editing technologies that are based on synthetic lambda integrases, allowing for specific insertion of large gene cassettes into the human genome, with an end goal to develop an iPSC-derived allogenic immunotherapy platform that can be broadly applied. This project will be part financed by a EUREKA GlobalStars competitive grant worth £400k.
In the latter half of 2023, Plasticell announced that they had secured funding for their project in collaboration with Cell and Gene Therapy Catapult, an independent innovation and technology organisation who specialise in the advancement of cell and gene therapies, and Imperial College London. Innovate UK Biomedical Catalyst awarded them £800k of funding to help them develop an integrated and scalable platform able to manufacture allogeneic iPSC derived immunotherapies.
T-Therapeutics
T-Therapeutics, a University of Cambridge spinout, is a next-generation T cell receptor (TCR) company. They were founded in 2022 by CEO Professor Allan Bradley, who is a professor in the University of Cambridge's Department of Medicine and is well-known for his role in the development of ES cell technology while he was a graduate student at Cambridge. This work was acknowledged when his thesis advisor, Martin Evan, received a 2007 Nobel Prize.
This company was founded with the mission to create safe, effective treatments for different types of cancer and autoimmune diseases by harnessing the power of T cell biology. T cells, which are a type of white blood cells, play an important role in the immune system, specifically for immunity of foreign substances by circulating until they find their specific antigen and attack it. The TCR is the part of the T cell that binds to a foreign antigen or cancer neoantigen, causing the activation, proliferation, and differentiation of the T cell. By utilising everything explained here, T-Therapeutics is developing drugs that can identify the cancer specific self-antigens and direct T cells to exterminate these cancer cells.
Their proprietary OpTiMusTM platform, which is based on a fully humanised TCR mouse able to provide a nearly unlimited source of unique, antigen-specific human TCRs, is how they are developing optimal TCR based therapeutics. This platform is a highly engineered transgenic mouse that carries genes for the human immune system. By immunising the mouse with human peptides, it is able to make humanised TCRs that do not exist in the human repertoire, providing an almost unlimited supply of TCRs with unique properties such as diversity, activity, half-life, and affinity.
Late-2023, T-Therapeutics had a stand-out achievement by raising £48m in a Series A financing, which will be used to discover and develop novel TCR therapeutics for cancer indications and inflammatory disorders. This financing was led by Sofinnova Partners, F-Prime Capital, Digitalis Ventures, and Cambridge Innovation Capital (CIC), along with participation from Sanofi Ventures and the University of Cambridge Venture Fund.
EnteroBiotix

Based in Aberdeen, Scotland, EnteroBiotix's main mission is to transform the lives of patients by providing microbiome medicines. Incorporating in 2017 and officially beginning operation in 2018, the company was founded by the CEO Dr James McIlroy, a qualified medical doctor and biopharmaceutical entrepreneur, who is also an inventor with his name on several microbiome related patent applications.
Their full-spectrum therapeutics encapsulate every beneficial component that are naturally found in a healthy gut microbiome, but in a dried, colourless, and odourless powder. Their lead product is EBX-102, an orally administered capsule containing the previously mentioned powder. It is a superior approach of preventing and treating disease by restoring and enhancing gut microbial ecology, compared to intestinal microbiota transfer (IMT).
In the beginning half of 2023, they announced the start of a Phase II clinical trial that will evaluate EBX-102 for the treatment of liver cirrhosis and hepatic encephalopathy. This study, which has been called Intestinal Microbiota Product in Liver Cirrhosis and Encephalopathy (IMPuLCE), will enrol 56 patients with liver cirrhosis across UK sites.
Applied Genomics Limited

Based in Norfolk, Applied Genomics are providers of environmental DNA (eDNA) analysis services. Founded by Ian Wilson in 2014, Ian is a successful innovator and boundary pusher, having found six other successful companies in the field. Most notably, he is the founder of Benthic Solutions, a marine environmental consultancy and survey company that has revolutionised methodologies in the UK sector. His companies offer specialised and unique services worldwide, thriving even during challenging times, such as during the COVID-19 pandemic.
Applied Genomics' mission, from incorporation to now, is to provide an alternative to taxonomy, which is the science of naming, describing, and classifying all organisms. They recognised that there was an unsustainable element within their current business model for marine surveys as their taxonomy department would not be able to support the growth they expected, and so they developed an alternative using eDNA, which is DNA that has come off an animal or plant as part of its life cycle. They also recognised that this alternative method could be used for other areas, including water quality, pollution monitoring, and looking for fisheries and for invasive species. Because of this, they find themselves working on very diverse projects, including those focused on air quality, water quality, sediment quality, biodiversity profiling, and regulatory requirements, such as invasive species. Currently, they are working on a project where they are trying to indicate microbial source tracking. In other words, they are attempting to answer the question of, "if there is pollution present in a river, where is it coming from and what kind of animal is the source?"
By supplying eDNA analysis services for areas such as biodiversity monitoring, environmental impact assessment, biosecurity surveillance, and threatened species detection, they are building a more comprehensive understanding of where the 'wild things' are. Their solutions are designed to work for local all the way to landscape-scale assessment no matter the environment. Their cost-effective and non-invasive approach provides ecological assessments at a molecular accuracy quickly and with significantly improved delivery.
Their core services can be split into three categories:
- Detection: Their provision of sample collection kits and analysis for targeted detection of species eDNA from environmental samples, allows for the sensitive and accurate detection of targeted species. In order to maximise the probability of eDNA capture and detection, they have developed large-volume sample methods.
- Monitoring: They analyse eDNA samples providing a comprehensive surveillance of all organisms in the surveyed environment that can be detected by eDNA. This biodiversity assessment is helpful in identifying sensitive receptors, assisting in modelling effect zones by providing needed data, and characterising indicator taxa, where the parameters (density, presence or absence, or infant survivorship) of either species or higher taxonomic groups are used as proxy measures of ecosystem conditions, in the presence of environmental pressures.
- Management: They are able to deliver data-driven insights, supporting clients' adaptive environmental management decisions, site-based environmental impact analyses, mapping of population genetic diversity and informing valuations of natural capital.
An example of what their methodology is can be taken from a water sample. They can extract the organic material from a water sample, and then extract the DNA from this material. By amplifying the DNA, a sequencer is able to analyse it and so the species that the DNA belongs to is identified. They can have between 20-30,000 DNA reads, with each read being a gene sequence of an animal. With a very complex computer program, they can identify which species from that read. This is also used for identifying pollution, as if they pick up a big signature from say human or cow, they can deduce that the faeces has not come from any animal in the water and has been washed off from the land.
Applied Genomics took a major step forward recently. They have developed a machine learning model to replicate the impact seen around a fish farm. This is to satisfy a regulatory requirement, as fish farms have to be monitored on every biomass cycle, which is about every 18 months, to prove that they are not having an impact beyond their planning license. Traditionally, this monitoring is done by more older techniques, such as marine macroinvertebrate surveys, which are slow, expensive, and require many taxonomists. They first took this alternative method to SEPA (Scottish Environmental Protection Agency) about five years ago. SEPA has become the first regulator to accept eDNA as an alternative to other methodological techniques for marine observation. Now, Applied Genomics have a regulator approved tool; the only company in the country to be involved in this and offer this service. They are hopeful to roll this out across the industry later in the year, alongside their continuous goal of expanding their company, their services, and their work.
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.