- within Tax, Energy and Natural Resources and International Law topic(s)
- with readers working within the Insurance industries
On 2 April 2019, the European Union ("EU") requested initiated a formal dispute before the World Trade Organization ("WTO") against Turkey's program to localize the production of the pharmaceutical products in Turkey. The consultation request focuses on the measures adopted by the Turkish government in 2016 to promote the local production of pharmaceutical products, which are otherwise imported into the country. The EU argues that the measures adopted Turkey are in contradiction with its obligations under the General Agreement on Tariffs and Trade ("GATT"), the Agreement on Trade Related Investment Measures ("TRIMs"), the Agreement on Trade – Related Aspects of Intellectual Property Rights ("TRIPS") and the Agreement on Subsidies and Countervailing Measures ("SCM"). Subject to the outcome of the procedural consultations between the EU and Turkey under the rules-based system of the WTO, the disputes seems to be evolving into a litigation phase. Considering that a complaint by the EU against Turkey would be rare (if not the first) in the enforcement history of the WTO era, the upcoming procedures are closely followed by the interested parties. Having its impact on the ever-important pharmaceutical industry, the outcome of the dispute may require Turkey to undertake some fundamental revisions in its localization measures to comply with its international obligations. Against this backdrop, this article reviews the circumstances surrounding the EU's complaint along with the sensitive nerve-endings in the Turkish pharmaceutical system, which are brought under the review of the WTO dispute settlement regime.
Structure of Turkish pharmaceutical market
The primary legislation regulating human medicinal products is the Law No. 1262, which is implemented via the Regulation on Classification. The scope of this legislation covers natural or synthetic origin active substance or combination of substances administered to human beings for the purposes of treating or preventing a disease, making a diagnosis, correcting, regulating or modifying a physiological function. The main players of the Turkish medicinal markets are the producers and importers that supply the needs of the market along with the pharmaceutical warehouses and pharmacy stores that conduct the wholesale and retail sale of medicinal products, respectively. In the administrative front, the Agency mainly regulates the licensing affairs and the Social Securities Institution ("SSI") maintains the reimbursement functions, while the Ministry of Health holds general governance and supervision responsibilities.
There are separate licensing requirements for the undertakings operating in the production and supply of medicinal products. Such requirements are administrated by the Licensing Regulation, according to which human medicinal products cannot be released to the market before the required licenses are obtained. The administrative organ authorized for examining applications and issuing licenses regarding production, importation and sale of human medicinal products is the Agency. Licenses are valid until they are suspended or cancelled. In case the license for a certain human medicinal product is suspended or cancelled, production or importation of the product in question shall cease. The list of license cancellations is published by the Agency on an annual basis. At times of suspension or cancellation, it falls within the responsibilities of the Ministry of Health to decide on products, which are already placed on the market. Another function of the Ministry of Health is reviewing applications by license holders to assign their licenses to other real persons or legal entities, which is allowed in the Licensing Regulation.
With respect to the distribution activities, there is a separate licensing requirement. By virtue of the Regulation on Pharmaceutical Warehouses, wholesale of human medicinal products is conducted by licensed pharmaceutical warehouses. The same legislation also prohibits the distribution of unlicensed, imitated or defected products. In case of a failure to comply with this prohibition, the said products shall be withdrawn from the market in accordance with the Withdrawal Regulation.
In the payments and reimbursement front, a reference pricing system is followed in Turkey. Based on this system, the pricing of medicinal products are determined by the acceptance of the lowest ex-factory price to the warehouses in certain reference countries such as France, Italy, Spain, Portugal and Greece. Accordingly, the payments for such products are made in accordance with the reimbursement system for pharmaceuticals which stipulates a maximum of 10% above the lowest priced pharmaceuticals in the generic groups. Discounts may separately be applied by medicinal companies and pharmacy stores in public pharmaceutical purchases.
Accordingly, alternative procurement processes are regulated under the procurement processes foreseen in the SSI Regulation Concerning the General Health Insurance Alternative Reimbursement ("Regulation"). Based on this legislation, the procurement process is initiated by an application to be made to the SSI. The application might be made by companies or via administrative bodies. The applications are then examined by the relevant General Directorates within the SSI in accordance with the Regulation. These General Directorates make the initial technical and economic evaluations concerning the application, prepare draft reimbursement models and contracts to be submitted to the relevant divisions. In practice, the General Directorates may also include the relevant companies in the process by inviting their representatives to meetings. Subsequently, the relevant division would determine the final reimbursement models, the draft contracts and the price to be paid for the relevant health services, which would incentivize domestic production and increase exports. After such determination, the companies that satisfy the preliminary qualifications are singled out and invited to submit their bids. In the selection process, the relevant division is required by the Regulation to not only take into consideration the price but also other parameters such as quality, efficiency and technical superiority. Once process is consummated, the contract is signed by the representative of the company and the president of the SSI. The Regulation states that the relevant General Directorates would oversee implementation of the contract.
Turkish pharmaceutical market in numbers
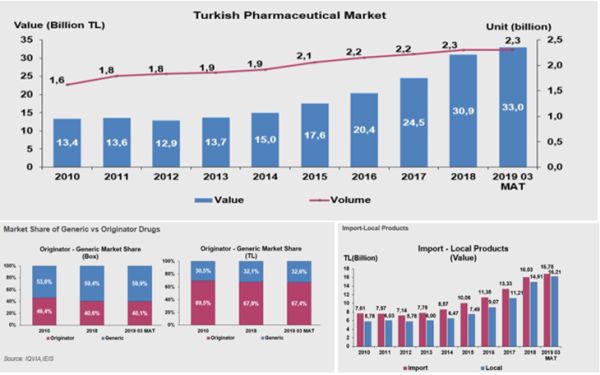
Based on the data provided by the Pharmaceutical Manufacturers Association of Turkey, the market size of pharmaceuticals in Turkey has reached TL 33 billion (approximately USD 5.5 billion) for the last 12 months as of March 2019, while unit sales reached 2,3 billion units.
With respect to the product breakdown, oncology products has the largest share by value, which amounts to 12,8% of the total market. In terms of volume, antibiotic products and anti-rheumatism products represents the largest share in the total market, which amounts to 11.1%. When we focus on exports, pharmaceutical exportation in Turkey has reportedly reached USD 1173 million in the last 12 months as of March 2019. In a statement last year, the Minister of Health provided that local production amounts to 80% (in terms of units) of the total pharmaceuticals consumed in Turkey. However, the Minister adds that local production only amounts to 50% in terms of value. 1
Below are some key indicators of the market which provides a general overview as well as the breakdown between original-generic products and imported-local products extracted from the records of the Pharmaceutical Manufacturers Association of Turkey2:
Turkey's measures subject to the EU complaint
The measures subject to the EU's complaint to the WTO are mainly regulated within the framework of an Industry Cooperation Program in Health Services ("HICP"). The Communique on Applying Industry Cooperation Program in Health Service Areas ("Communique"), which is the to go legislation for the governance of the measures under scrutiny by the EU aims to incentivize domestic production of certain goods and services in the health sector that are being imported. The Communique provides a set of rules and principles where undertakings operating in the Turkish market for pharmaceutical products are imposed with certain responsibilities to cooperate with the relevant administrative bodies and to increase the domestic production. Accordingly, the overall cooperation mechanism entails a number of commitments imposed on relevant the undertakings, which are brought under the review of the WTO. To that end, there are four categories of commitments (A, B, C and D), which are explained below.
Category A (Local Support Contribution) - The contractor undertakes to produce high tech goods or its relevant parts by using existing production facilities and local industry abilities in Turkey (detailed information may be found on Annex – 1 of the Regulation). Category A commitments can be fulfilled through a partnership or a subcontractor. The Contractor may directly contribute to the joint production within the scope of Health ICP by using existing facilities and abilities of the producer. The Contractor may also use a domestic subcontractor regarding the production of aforementioned goods or services.
Category B (Investment) - The contractor undertakes to produce high tech goods or its relevant parts by a new production facility to be established (detailed information may be found on Annex – 2 of the Regulation). Category B refers to investments to be made by the Contractor. Commitments required in this category can be fulfilled through establishing a new production facility or by purchasing an existing facility. The Contractor may build a new facility for the production of the aforementioned goods and services within the scope of Health ICP. The Contractor may also purchase an existing facility and carry production activities.
Category C (Technological Cooperation) - The contractor undertakes to bring technological capabilities in the local industry. These obligations shall be fulfilled in guidance with activities referred to in Annex – 3 (detailed information may be found on Annex - 3 of the Regulation).
Category D (Export) - The Contractor undertakes to execute export activities in Turkey. These obligations shall be fulfilled in guidance with activities referred to in Annex 4 (detailed information may be found on Annex – 4 of the Regulation).
It should be noted that commitments within the context of Category C or Category D shall only be given together with Category A and/or Category B commitments. Against this backdrop, Hence, it will be prudent to break down the enforcement trends of the foregoing localization measures and have a grasp on the main points of objections by the EU.
Measures under review
In light of the foregoing, the EU's consultation request focus on certain measures adopted by Turkey as part of its centralized efforts to localize the production of pharmaceutical products in consumption. Overall, the EU argues that the implementation of the measures within the framework explained above rise discriminatory measures concerning trade-related investments. Accordingly, in essence, the EU's objections are concentrated on the following four Turkish measures.

Localization of production - With respect to the claims over the localization requirement, which is the main issue at hand, the EU argues that Turkey adopted comprehensive policies to localize the production of pharmaceutical products' consumed in Turkey. The EU argues that Turkey requires foreign producers to commit to localise their production in Turkey. In case of a failure to localize, the Turkish authorities are alleged to exclude the relevant enterprises from the social security reimbursement system. In the Turkish pharmaceutical landscape, the social security reimbursement scheme includes the majority of products in consumption. Therefore, the exclusion of the pharmaceutical companies who have failed to localize their productions from the fundamental reimbursement channel rises an anti-competitive barrier to foreign producers when compared to the domestic producers in Turkey. In other words, the EU claims that the reimbursement scheme of social security system constitutes a subsidy and the requirement to use domestic goods over imported goods in the localization requirements amounts to a prohibited subsidy. In addition, the EU also argues that measures are applied in a non-transparent manner and may differ from producer to producer.

Technology transfer requirement - As part of the localization requirement, the EU also claims that Turkey requires foreign producers to transfer the details of their technologies and use them through a producer established in Turkey. This allegation indicates that such technology transfer requirement include the patent rights. Accordingly, the EU argues that Turkey does not provide the same protection to EU derived intellectual rights when compared to the domestically derived intellectual property rights and the use of foreign patent rights subsequently face a discrimination. Furthermore, the EU stipulates in its consultation request that the restriction on the use of patent rights (such as sub-licensing) lead to the disclosure of confidential information pertaining to the foreign producers.
Import ban on localized products - The EU alleges that once the production of a given pharmaceutical product has been localised in Turkey, the import of that pharmaceutical product becomes prohibited. In other words, the claims stipulate that Turkey not only pressure the foreign producers to localize their production but also restricts import to solidify the now-localized production.
Prioritization measure - The EU argues that in certain cases, where the imported products are not excluded from the reimbursement scheme, the Turkish authorities prioritize the applications of the domestic products to be included into the system over the imported products. Under this discrimination claim, the EU further argues that that the operations surrounding the system are not handled in a transparent manner. The application of measures are not uniform, impartial or reasonable,
Conclusion
The request for consultation initiates a formal dispute procedure between the parties before the WTO. If the parties cannot reach a mutually satisfactory solution at the end of the 60 days consultation, the EU may request the establishment of a panel, which will subsequently resolve the dispute. In a similar process dated 8 March 2017, Turkey requested a consultation with the United States ("US") for the countervailing measures applied to pipes and tubes. Upon failure of this consultation, Turkey requested the establishment of a panel, which has subsequently ruled that the relevant applications of the US were in violation of its obligations under the SCM.
The measures listed in the consultation request concern the production, import and marketing pharmaceutical products in Turkey. Therefore, every enterprise operating in the pharmaceutical sector may be effected by the outcome of the dispute at hand. As such, if the consultation leads to a litigation and the WTO adjudicating bodies rule in favor of the EU's arguments, Turkey will have to bring the relevant measures into conformity with its international obligations. In such a case, all the companies that operate or invest in the Turkish pharmaceutical sector will have to adopt their business plans, productions and supply chains in accordance with the new landscape.
Footnotes
1. https://www.turkiyegazetesi.com.tr/saglik/553740.aspx
2. http://www.ieis.org.tr/ieis/en/indicators/33/turkish-pharmaceutical-market
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.


