- within Food, Drugs, Healthcare and Life Sciences topic(s)
- within Criminal Law, Cannabis & Hemp and Immigration topic(s)
On May 6, 2024, following more than a decade of discourse with interested stakeholders on potential approaches to regulation of laboratory developed tests (LDTs), the U.S. Food and Drug Administration (FDA) published its final rule setting forth its framework for oversight of LDTs. The final rule and accompanying policy to phase out the agency's general policy of "enforcement discretion" for LDTs comes roughly six months after FDA published its proposed rule that outlined the agency's proposed approach to increasing oversight over LDTs.
As detailed in our prior analyses of the proposed rule (see here and here), FDA proposed to implement a phaseout policy that would, across five stages and within four years, apply to clinical laboratories offering tests as LDTs the same regulatory requirements applicable to in vitro diagnostics (IVDs). The proposed rule received more than 6,500 comments, and while FDA did not change its amendments to the regulation or meaningfully modify the phaseout timeline, FDA has significantly modified its phaseout policy to extend full or partial enforcement discretion to additional categories of LDTs, creating a framework whereby the agency intends to take a more targeted enforcement approach, particularly in the near-term, to addressing LDTs.
In this Insight, we highlight the key features of the final rule and five-stage phaseout policy.
What Changes Were Made to FDA's Regulations?
In the final rule, FDA finalized its proposal to make a small but significant change to its regulations that reflects the agency's stance that LDTs are "devices" as defined under the Federal Food, Drug, and Cosmetic Act (FDCA) and are therefore subject to FDA's authority. Specifically, the final rule adds only eight words into FDA's regulation defining "in vitro diagnostic products" (21 C.F.R. § 809.3(a)) to make explicit its interpretation that IVDs are "devices" as defined in Section 201(h)(1) of the FDCA, even when "the manufacturer of these products is a laboratory."
The final rule is effective on July 5, 2024.
The remainder of the 160-page final rule, as published in the Federal Register, is dedicated to describing FDA's five-stage phaseout policy as well as addressing the numerous public comments received from stakeholders. The public comments are addressed in 307 separate responses and cover, among other topics, the need for the rule, the legal authority for the rule and other legal comments, the impact of the rule and updated policy on small businesses, pricing, access and innovation, and the details of the final phaseout policy.
What Tests are Subject to FDA's Final Phaseout Policy?
Consistent with the proposed phaseout policy, FDA's final phaseout policy applies to "IVDs offered as LDTs," which comprise a broader set of tests than FDA's traditional understanding of an LDT.
| Tests Subject to FDA's Final Phaseout Policy | |
|---|---|
|
"IVDs offered as LDTs" |
"IVDs that are manufactured and offered as LDTs" by high-complexity laboratories that are certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) and "used within such laboratories," whether or not they fall within FDA's "traditional understanding of an LDT." |
|
"LDTs" |
FDA has generally considered an LDT to be "an IVD that is intended for clinical use and that is designed, manufactured, and used within a single" high-complexity laboratory certified under CLIA. |
Conversely, consistent with the proposed phaseout policy, the final
rule makes clear that certain tests are excluded from the final
phaseout policy and remain subject to active regulation since they
were never considered to qualify for FDA's general enforcement
discretion approach for LDTs.
| Tests that Remain Under Active Oversight | |
|---|---|
|
Test Type |
Description |
|
Direct-to-Consumer (DTC) Tests |
Tests intended for "consumer use (without meaningful involvement by a licensed healthcare professional)." |
|
Tests Intended as Blood Donor Screening or HCT/P Donor Screening Tests |
Tests intended as blood donor screening or human cells, tissues, and cellular and tissue based products (HCT/Ps) donor screening tests required for infectious disease testing or for determination of blood group and Rh factors. |
|
Tests Intended for Emergency Use |
Tests intended for emergencies, potential emergencies, or material threats declared under Section 564 of the FDCA. |
For tests intended for emergency use, FDA has generally expected
emergency use authorizations (EUAs) for LDTs after a declared
emergency. However, FDA has issued a draft guidance entitled
"Consideration of Enforcement Policies for Tests
During a Section 564 Declared Emergency" (May 2024) on
factors to consider in adopting enforcement discretion policies
relating to uses of unauthorized tests in future public health
emergencies. Further, FDA has issued a draft guidance entitled
"Enforcement Policy for Certain In Vitro Diagnostic
Devices for Immediate Public Health Response in the Absence of a
Declaration under Section 564" that proposes a new
enforcement discretion policy for certain "immediate
response" tests manufactured and offered by laboratory
manufacturers in the absence of a declared emergency under Section
564 of the FDCA.
Last but not least, FDA clarifies in the preamble to the final rule that LDTs offered for a use that is the same as a companion diagnostic (CDx), including those LDTs described in FDA's "Oncology Drug Products Used with Certain In Vitro Diagnostics Pilot Program," are subject to the final phaseout policy that generally applies to "IVDs offered as LDTs."
What Tests Will Generally Remain Under Full Enforcement Discretion?
The final phaseout policy confirms that FDA will continue exercising full enforcement discretion (i.e., generally not enforce all applicable FDA requirements) for the four types of tests originally described in the proposed rule as well as LDTs manufactured and performed within the Veterans Health Administration (VHA) or the Department of Defense (DoD).
| Tests Generally Remaining Under Full Enforcement Discretion | |
|---|---|
|
Test Type |
Description |
|
"1976-type" LDTs |
Tests that (1) involve use of manual techniques (without automation) performed by laboratory personnel with specialized expertise, (2) use components legally marketed for clinical use, and (3) meet FDA's traditional understanding of an LDT. |
|
Human Leukocyte Antigen (HLA) Tests for Transplantation |
HLA tests designed, manufactured, and used by a single laboratory certified under CLIA that meet the requirements to perform high-complexity histocompatibility testing when used in connection with organ, stem cell, and tissue transplantation to perform HLA allele typing, for HLA antibody screening and monitoring, or for conducting real and "virtual" HLA crossmatch tests. |
|
Tests for Forensic (Law Enforcement) Purposes |
FDA maintains that it has a "longstanding enforcement discretion approach" for tests intended solely for forensic (law enforcement) purposes, irrespective of whether such tests are offered as an LDT. |
|
Tests Used for Public Health Surveillance |
FDA maintains that tests exclusively used for public health surveillance are distinct where (1) they are intended solely for use on systematically collected samples for analysis and interpretation of health data in connection with disease prevention and control and (2) test results are not reported to patients or their healthcare providers. |
|
VHA and DoD LDTs |
LDTs manufactured and performed within the VHA or DoD. This policy applies only to "LDTs used for patients that are being tested and treated within the DoD or VHA." |
What Tests Will Generally Remain Under Partial Enforcement
Discretion?
The final rule has added a number of significant categories of tests that will remain under some degree of enforcement discretion. Specifically, FDA is adopting several new "targeted enforcement discretion policies" for the following.
| Tests Generally Remaining Under Partial Enforcement Discretion Discretion | ||
|---|---|---|
|
Test Type |
Description |
Enforcement Discretion Limited to the Following Requirements |
|
Same Healthcare System/Unmet Need LDTs |
LDTs manufactured and performed by a laboratory integrated within a healthcare system to meet an unmet need of patients receiving care within the same healthcare system. |
Premarket review requirements Quality System (QS) requirements (except for recordkeeping requirements) |
|
Currently Marketed "IVDs Offered As LDTs" |
"IVDs offered as LDTs" that were first marketed prior to the date of issuance of the final rule and that are not modified or that are modified in certain limited ways. |
Premarket review requirements QS requirements (except for recordkeeping requirements) |
|
Blood Establishment Non-Molecular Antisera LDTs for Rare RBC Antigens |
Non-molecular antisera LDTs for rare red blood cell (RBC) antigens where such tests are manufactured and performed in blood establishments, including transfusion services and immunohematology laboratories and where there is no alternative available to meet the patient's need for a compatible blood transfusion. |
Premarket review requirements QS requirements (except for recordkeeping requirements) |
|
NYS CLEP LDTs |
LDTs that are approved, conditionally approved, or within an approved exemption from full technical documentation, under the New York State Department of Health Clinical Laboratory Evaluation Program (NYS CLEP) |
Premarket review requirements |
|
Modifications to Certain FDA-Authorized IVDs |
Modifications made by a high-complexity laboratory certified under CLIA to another manufacturer's 510(k)-cleared or de novo authorized IVD, following design controls and other applicable QS requirements, in a manner that could not significantly affect the safety or effectiveness of the test and does not constitute a major change or modification in intended use, and where the modified test is performed only in the laboratory making the modification. |
Premarket review requirements |
What is the Timeline for the Final Phaseout Policy?
FDA plans to phase in the various regulatory requirements for "IVDs offered as LDTs" that do not fall under a continued enforcement discretion policy as described above across five stages and within four years as outlined below.
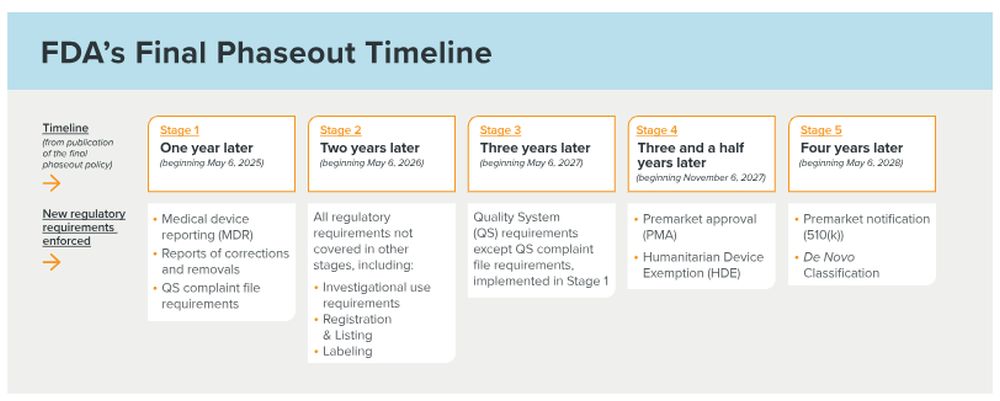
Stage 1: One year after publication of the final rule, FDA will phase in medical device reporting (MDR) requirements, reports of correction and removal requirements, and QS complaint file requirements.
Stage 2: Two years after publication of the final rule, FDA will phase in all other medical device requirements not covered in the phaseout policy, including investigational use requirements, registration and listing requirements, and labeling requirements.
Stage 3: Three years after publication of the final rule, FDA will phase in the QS requirements in 21 C.F.R. Part 820 (excluding the QS complaint file requirements, which are included in Stage 1).
For LDTs (i.e., when all manufacturing activities occur within a single laboratory and the test is not transferred outside that laboratory), this is limited to the phase in of requirements covering design controls, purchasing controls (including supplier controls), acceptance activities (receiving, in-process, and finished device acceptance), corrective and preventive action (CAPA), and records requirements.
Stage 4: Three and a half years after publication of the final rule, FDA will phase in the premarket review requirement for high-risk "IVDs offered as LDTs" and will begin enforcing the premarket approval (PMA) requirement.
FDA has announced its intention to complete reclassification of certain Class III IVDs to Class II IVDs in advance of this deadline.
Stage 5: Four years after publication of the final rule, FDA will phase in the premarket review requirement for moderate-risk and low-risk "IVDs offered as LDTs" (that require premarket submissions) and will begin enforcing the premarket notification (510(k)) and de novo request for classification requirements.
With respect to the premarket review requirements in Stages 4 and 5, FDA does not generally intend to enforce against "IVDs offered as LDTs" for lacking premarket clearance or approval if an appropriate premarket submission is received by the beginning of the applicable stage. Instead, FDA intends to continue exercising enforcement discretion for the pendency of the agency's review of the submission.
What's Next?
It is anticipated that laboratory associations and individual laboratories will consider pursuing legal action against FDA and seek to enjoin the agency from implementing the final rule. Further, it remains to be seen whether the issuance of the final rule will spur further discussions of legislative approaches to regulating LDTs.
While the final rule has created several new and notable categories for ongoing enforcement discretion for LDTs, it is notable that such ongoing enforcement discretion is not guaranteed. As stated (and reiterated) in the final rule:
"... regardless of this or any other enforcement discretion policy, FDA retains discretion to pursue enforcement action at any time against violative IVDs when appropriate. We intend to carefully monitor LDTs falling within this policy and intend to take action regarding such LDTs as appropriate taking into account any public health concerns as evaluated on a case-by-case basis."
Indeed, as stated in the final rule, FDA intends to "take targeted steps" to address Currently Marketed IVDs Offered as LDTs that are problematic, including those that the agency views to be potentially inaccurate or poorly validated. As part of this initiative, FDA states that it intends to, by May 6, 2026, collect labeling from laboratories offering Currently Marketed IVDs Offered as LDTs. Further, FDA states that the agency "generally intends to take action where the labeling of a currently marketed IVD offered as an LDT is false or misleading, and/or the IVD offered as an LDT lacks the appropriate assurance of safety and effectiveness for its intended uses as a result of any such claims that are not adequately substantiated." FDA similarly intends to request labeling for NYS CLEP LDTs and Same Healthcare System/Unmet Need LDTs.
Therefore, it is important for laboratories developing and offering LDTs to carefully consider FDA's new framework for oversight and to appropriately adjust their regulatory strategy and compliance plans.
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.




